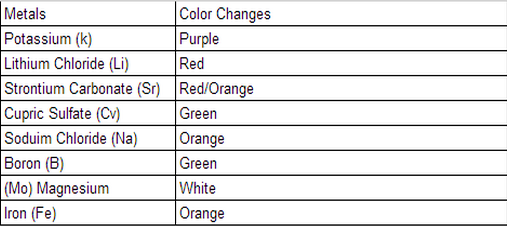
| My group partners and I performed a scripted lab provided by our chemistry teacher. This is my favorite lab so far. We used 6 different elements and burned them using a bunsen burner to note what color the elements produced. The elements are potassium, magnesium, sodium chloride, lithium chloride, cupric/copper sulfate, and strontium. We would use the elements one-by-one with a drop of alcohol and put it over the bunsen burner flames. I was very excited to do this lab and didn't know what to expect. Below is a video of some of the elements we recorded during the lab. |
| It was very exciting to see the different colors the elements produced. My group and I learned that scientists use flame test labs to determine what unknown substances are since they all give off a different energy when given more energy, like heat. The wavelengths of energy matches particularly with the wavelengths of light and that's why the colors are produced. Because certain elements give off certain colors, that's how scientists can determine what substances are made of. After looking at the graph, that is also attached below, I noticed a red/orange repetition for some elements, which was interesting. Since they categorize within the same color wavelength, I wonder what substances those elements have in common. Overall, I think the lab went very well. We completed the lab with ease and no burns. One thing I would like to change would be to do all of the tests with the lights off so we could have an easier time to see the energy given off. But some elements were easy to see even with the lights on, which I was very thankful for. I would be open to doing this lab again with different elements, or even creating my own element. |